If you want a detailed guide on the covalent bonding of water (H2O), you will love this amazing guide I am about to share with you.
But first of all, let’s start with a simple introduction.
In Chemistry, a covalent bond is formed due to the sharing of electrons between atoms. But, why does this “sharing of electrons” take place?
Let me explain. But before that, let’s talk about covalent bonding in detail.
What is Covalent Bonding?
In this type of bonding, the “participating” atoms share electrons with each other to become stable.
Here is what I mean.
In an outermost shell, an atom should have two or eight electrons to achieve the atomic configuration of a noble gas.
Noble gases are unreactive (as they have fully-filled outer shells). So, whenever an atom becomes stable, it is said to have achieved the electronic configuration of a noble gas.
Pretty simple, isn’t it?
Important Note: At this stage, you might be wondering, why not atoms exchange electrons like ionic bonding? Why does this exchange of electrons take place?
The answer is simple, ionisation energy (energy essential to take away an electron).
Elements that have high ionisation energy cannot transfer electrons. Plus, the elements having a low electron affinity cannot take up electrons.
As a result, the atoms of such elements share electrons with each other (to achieve stability).
The two ways in which covalent bonding can be achieved:
- The sharing of electrons between atoms of different elements (for example H2O, CH4 and NH3 etc).
- The sharing of electrons between atoms of the same elements (for example N2, H2 and O2 etc).
If I talk about water (H2O), we will be talking about the first method.
Covalent Bonding of Water (H2O):
Here’s how it takes place.
If I talk about hydrogen (H1), it has one electron in its outermost shell. To become stable, the hydrogen atom needs one electron to achieve a duplet electronic configuration (the duplet rule).
The Hydrogen atom follows the duplet rule that says: An atom is stable if it has two electrons in its outer shell.
Moving on, the Oxygen atom (O8) has six electrons in its outer shell. Therefore, it needs two more electrons to become stable (achieve octet electronic configuration).
The Oxygen atom follows the octet rule in which the atom completes eight electrons in its valence (outermost) shell.
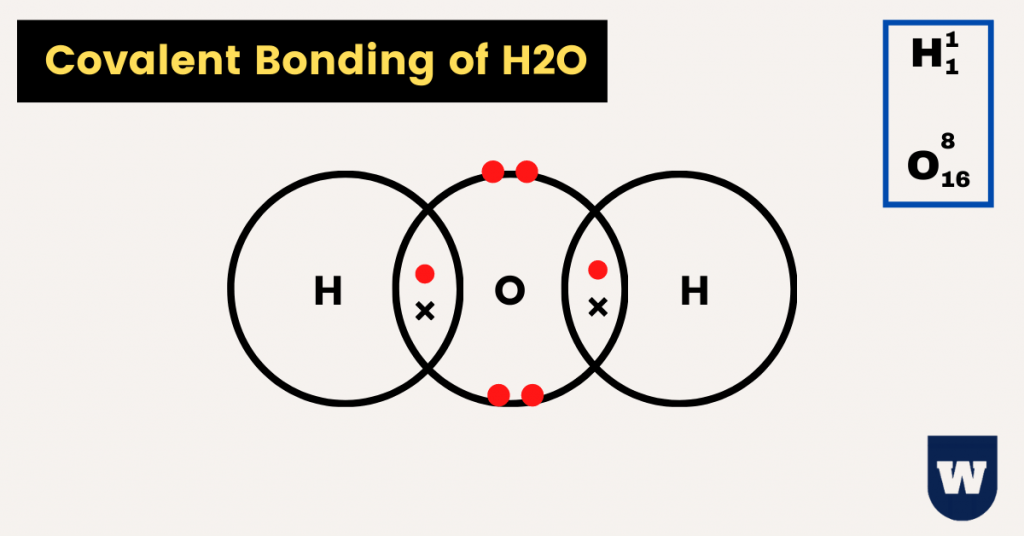
As you can see in the image above, two hydrogen atoms and one oxygen atom share electrons with each other to become stable.
Water is a polar covalent molecule. This is because of the unequal sharing of electrons (due to the difference in electronegativity) between oxygen and hydrogen.
In short, here is the summary.
- An oxygen atom has six electrons and needs two more electrons (according to the octet rule).
- A hydrogen atom has one electron and needs one more electron (according to the duplet rule).
- As a result, the oxygen atom shares electrons with 2 hydrogen atoms. This results in the formation of a water molecule (H2O).
To further understand this topic, let’s take a look at another example to understand this concept.
Example: Covalent Bonding of Nitrogen:
This example will help you understand this topic in a better way.
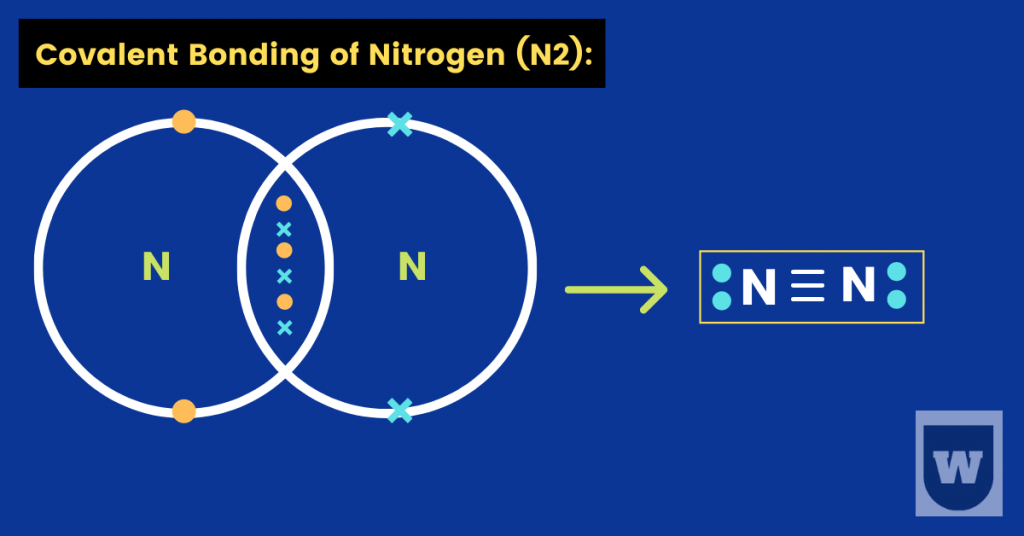
If I talk about the Nitrogen atom (N7), it has seven electrons in total (5 in its valence shell). This means that a nitrogen atom needs three more electrons to become stable.
As a result, two Nitrogen atoms share electrons with each other.
In simple words, a nitrogen atom can fill its octet by sharing three electrons with another nitrogen atom. This results in the formation of three covalent bonds, a so-called triple bond.
Pretty simple, isn’t it?
Further reading:
Ionic Bonding of sodium chloride (NaCl) | Best Guide
Electrolysis Made Simple | Notes
Exothermic and Endothermic reactions | Complete Guide
With this, it is time to move on and talk about the properties of covalent compounds.
Properties of covalent compounds:
Here are the four properties that you should know:
- Covalent bonding involves neutral molecules (having no electric charge).
This is the reason that forces of attraction between these molecules are weaker as compared to ionic compounds. Therefore, these compounds are usually volatile gases or liquids.
- Covalent compounds (generally) have low melting and boiling point.
Let’s recall from the first property.
As I mentioned above, these compounds are made up of neutral molecules. Plus, the forces of attraction between these molecules are weak.
So, a low amount of heat is required to break these bonds. This explains why they have a low melting and boiling point.
Note: Since the intermolecular forces are weak, covalent compounds are also soft and brittle (because they break or distort with force).
Now, let me tell you about the third property.
- Covalent compounds do not conduct electricity.
I have a question for you. What is the basic requirement to conduct electricity?
Ions or electrons.
And if you recall, covalent compounds involve neutral molecules. Since there are no free moving ions or electrons, these compounds do not conduct electricity.
In other words, these compounds do not have charged particles to transport electrons.
Before talking about the next property, let me tell you about polar and non-polar covalent bonds.
In Nonpolar covalent compounds, the electrons are shared equally between the two atoms. For example: In the covalent bonding of Hydrogen (H2), both hydrogen atoms share one electron with each other.
And, Nonpolar compounds are stronger.
On the other hand, polar compounds are different.
A polar compound is one in which the sharing of elections (or the attraction of electrons) is unequal.
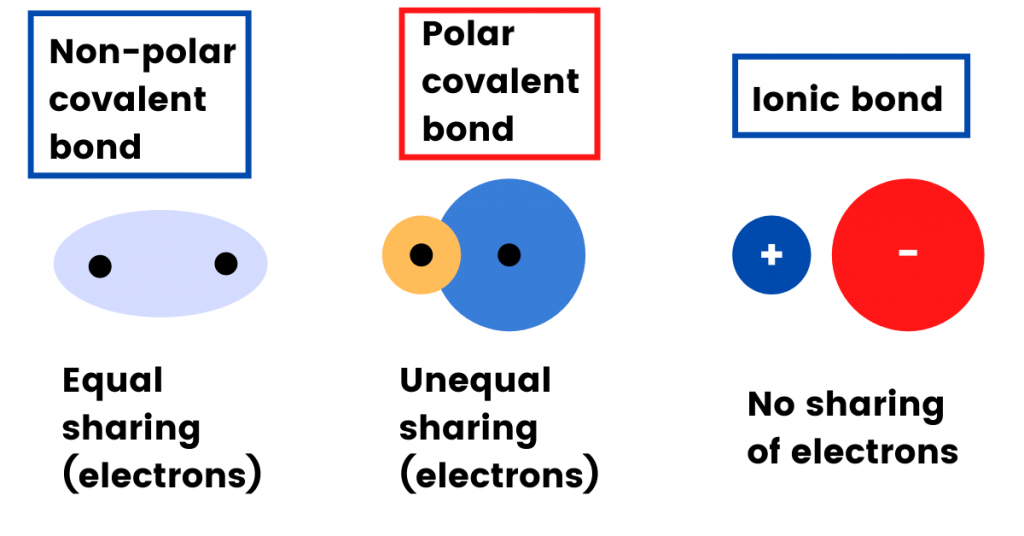
An example is the hydrogen chloride molecule (HCl). The bonding between hydrogen and chlorine leans more towards chlorine atoms.
This is because chlorine (Cl) is more electronegative in nature as compared to hydrogen.
- Covalent compounds are soluble in organic solvents but insoluble in water.
Here’s why.
Remember that most of the organic solvents are non-polar in nature. And, covalent compounds are also (mostly) non-polar.
Since “like dissolves like”, these compounds are soluble in organic solvents.
And, most of the covalent compounds are insoluble in water.
Since water is a polar solvent, it does not dissolve Nonpolar compounds. Plus, when covalent compounds are dissolved in water, they dissociate into molecules (but not into ions).
This takes us straight to our next topic, types of covalent bonds.
Types of Covalent Bonds:
The covalent bonds, based on the number of shared pairs of electrons, can be classified as:
- Single covalent bond
- Double covalent bond
- Triple covalent bond
Let’s take a look at each of them in detail.
Single bonds:
If only one pair of electron is shared between the two “participating” atoms, then we say that a single bond is formed.
In other words, only two electrons are shared in this type of bond.
Note: This is the most stable type of bond but has less density (and is also weaker) than double and triple bonds.
For example, hydrogen chloride (HCl).
A hydrogen atom has one valence electron while a chlorine atom has seven valence electrons. As a result, a single bond is formed between the hydrogen and chlorine atom by sharing one electron.
Double bond:
When two pairs of electrons are shared, we say that a double bond is formed. This type of bond is represented by two dashes (=).
As I mentioned above, this bond is much stronger but less stable than a single bond.
For example, carbon dioxide (CO2). The structural formula is: O=C=O
The carbon atom has four valence electrons while the oxygen atom has six valence electrons. So, each oxygen atom shares two electrons with two electrons on the central carbon atom.
So this forms a double bond.
Triple bond:
If three pairs of electrons are shared, we say that a triple bond is formed. This type of bond is represented by three dashes (≡).
You should know that these are the least stable types of covalent bonds.
An example is the Nitrogen molecule (N2).
A nitrogen atom has five valence electrons and it needs three more electrons to become stable. So in the formation of a nitrogen molecule, each nitrogen atom shares three electrons.
Therefore, a triple bond is formed.
Covalent Bonding vs Ionic Bonding:
When studying this topic, it is important to talk about this question as well.
Well, what is the difference between an ionic and covalent bond?
A covalent bond is formed between non-metals where sharing of electrons takes place. But in ionic bonding, cation (positive ion) and anion (negative ion) are involved where transfer of elections takes place.
Here is some detail for you.
An ionic bond is a type of chemical bond in which the atoms have different electronegativity values from each other. For example, sodium (Na) and chlorine (Cl) form an ionic bond to make NaCl (table salt).
However, in a covalent bond, the atoms are bound to share electrons. For example, if we talk about water (H2O), it is a polar covalent bond. This is because of the unequal sharing of electrons.
Here is a simple table for you to understand this topic in a better way:
| Ionic Bonds | Covalent Bonds | |
| Description | Transfer of electrons | Sharing of electrons |
| Polarity | High | Low |
| Melting & Boiling point | High | Low |
| State at room temperature | Solid | Liquid or Gas |
| Example | H2SO4, NaCl | H2O, N2 |
FAQ’s:
Here are some of the questions related to this topic.
Why is water covalently bonded?
Covalent bonding occurs between non-metals. Since both hydrogen and oxygen are non-metals, a covalent bond is formed between them.
This is suggested by the formula of water – H2O. This shows that two hydrogen atoms and one oxygen atom share electrons with each other to become stable.
How many covalent bonds are in H2O?
Two covalent bonds.
The concept is simple. The oxygen atom shares a pair of electrons with each hydrogen atom. Since there are two hydrogen atoms, two covalent bonds are formed that hold the water molecule together.
Wrapping up:
With this, our topic about the covalent bonding of water (H2O) has come to an end.
Now, it is your turn.
Which part of the topic do you find interesting? Is it related to drawing the structure of water (H2O), or is it about the difference between ionic and covalent bonds?
Either way, do let me know.
If you have any questions, you can drop them below. Thank You for staying with me till the end. Stay tuned for more.
